CE Expiration Date:
CEU (Continuing Education Unit): Credit(s)
AGD Code:
Educational aims and objectives
This clinical article aims to provide a review of endodontic-related nerve injuries using the literature.
Expected outcomes
Endodontic Practice US subscribers can answer the CE questions to earn 2 hours of CE from reading this article. Correctly answering the questions will demonstrate the reader can:
- Recognize the risk factors of post-endodontic neuropathy of the trigeminal nerve.
- Realize the incidence of endodontic nerve injuries.
- Realize the consequences of endodontic-related nerve injuries.
- Realize the mechanism of nerve injury related to endodontic treatment.
- Identify various aspects of related to neuropathic pain.
In the first of two articles, Dr. Tara Renton presents a literature review for the post-endodontic neuropathy of the trigeminal nerve
Sensory disturbances or peripheral neuropathies such as anesthesia, hypo-esthesia, hyperesthesia, and paresthesia may present in the trigeminal system due to many local and systemic factors. Spontaneous neuropathy must always be regarded with suspicion (red flag) and neoplasia excluded. However, if the neuropathy coincides with recent dental treatment, then exclusion of post-traumatic neuropathy must be excluded first.
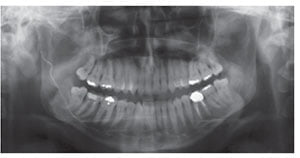
The inferior alveolar nerve (IAN) is at risk from a variety of dental procedures, in that the IAN is contained within a bony canal predisposing it to ischemia, trauma, and subsequent injury in relation to dental procedures. This may also result in a higher incidence of permanent damage for inferior alveolar nerve injuries compared with lingual nerve injuries (Pogrel, 2007). Causes of inferior alveolar nerve injury include local anesthetic injections, third molar surgery, implants, endodontics, ablative surgery, trauma, and orthognathic surgery.
Endodontists and dentists take for granted the rather unusual characteristics of the dental pulp (the only organ in health to display allodynia [pain] to all stimuli) and to respond innocuously to caustic high pH chemicals that would destroy and burn tissues elsewhere in the body. Thus, many compounds and chemicals routinely used in dentistry can cause severe irreversible tissue damage in structures close to the treated tooth.

Iatrogenic trigeminal nerve injuries remain a significant and complex clinical problem. Altered sensation and pain in the orofacial region may interfere with speaking, eating, kissing, shaving, applying makeup, tooth brushing, and drinking — in fact, just about every social interaction we take for granted. Thus, these injuries have a significant negative effect on the patient’s quality of life, and the iatrogenesis of these injuries leads to significant psychological effects (Smith, et al., 2013).
Incidence of endodontic nerve injuries
The most common nerve affected by endodontic procedures is the IAN (Alves, Coutinho, Gonçalves, 2014). Maxillary branches of the trigeminal nerve can also be damage-related endodontic treatment usually due to sodium hypochlorite leakage (Pelka, Petschelt, 2008) (Figure 1).
In one retrospective study examining cases of paresthesia related to endodontic treatment of mandibular premolars, the incidence was 0.96% (8/832) (Knowles, Jergenson, Howard, 2003). In a survey of 2,338 patients, 7% sustained chronic neuropathic pain after a single endodontic procedure (Klasser et al, 2011).
Most reports of endodontic-related nerve injuries are case reports with very little analysis of the clinical impact for the patient, risk factors, or management issues and are solely case reports.
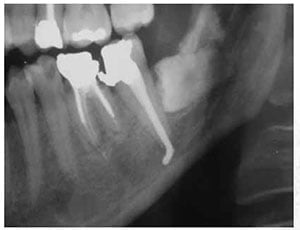
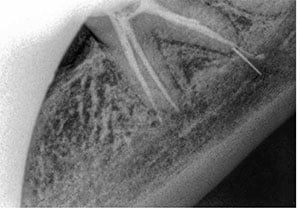
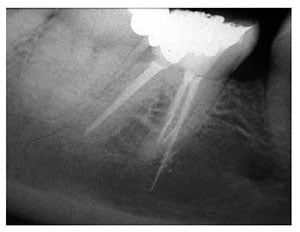
These case reports solely focus on over-instrumentation and loss of apical seal leading to endodontic material extrusion into the inferior dental canal (IDC), resulting in “mechanical” damage to the IAN (Figures 2-5). (Rowe, 1983; Yaltirik, Ozbas, Erisen, 2002; Gallas-Torreira, et al., 2003; Tilotta-Yasukawa, et al., 2006; Scolozzi, Lombardi, Jaques, 2004; Zmener, 2004; Vasilakis, Vasilakis, 2004; Poveda, et al., 2006; Pogrel, 2007; von Ohle, ElAyouti, 2010; Gonzalez-Martin, et al., 2010; Gambarini, et al., 2011; Marques, Gomes, 2011; Lopez-Lopez, et al., 2012; Orr, 1987; 1985; Dempf, Hausamen, 2000; Forman, Rood, 1977; Gallas-Torreira, et al., 2003; Tilotta-Yasukawa, 2006; Qrstravik, Brodin, Aas, 1983; Vasilakis, Vasilakis, 2004).
To date, there are four case series reports on inferior alveolar nerve injuries (IANIs) related to root canal treatment (RCT), including 61 cases (Pogrel, 2007), eight cases (Knowles, Jergenson, Howard, 2003), four cases (Scolozzi, Lombardi, Jaques, 2004), and one case series reported on the treatment of 11 cases (Grötz et al, 1998). These case series predominantly focus on the outcomes surgical exploration and irrigation of damage nerves.
Consequences of endodontic-related nerve injuries
The largest series of endodontic-related trigeminal nerve injuries in 61 patients over an 8-year period (Pogrel, 2007) reported that eight patients (12.5%) were asymptomatic, which is the only report to proactively highlight that overfilling and extrusion of endodontic materials into the IDC will not always cause neuropathy.
However, the majority of the patients reported symptoms (87.5%); 42 exhibited only mild symptoms (3-month-old injuries); 10 patients experienced some resolution; 11 patients were symptomatic and underwent surgical exploration — five of these within 48 hours and others up to 10 days to 3 months. (Four experienced partial resolution and two no recovery.) (Pogrel, 2007).
More recently, in a personal communication, Dr. Pogrel has undertaken a further eight cases as urgent cases (within 24 hours) and reports significantly improved resolution of neuropathy and pain in these patients.
Neuropathic pain
Persistent pain after endodontics has been reported to occur in 3%-13% of patients (Marbach, et al., 1982; Lobb, Zakariasen, McGrath, 1996) while surgical endodontics resulted in chronic neuropathic pain in 5% of patients (Campbell, Parks, Dodds, 1990).
In a previous study of 135 patients with IANI caused by dental treatment or malignancy, 22% presented with dysesthesia, which was significantly associated with the female gender (Caissie, et al., 2005). In another study, the significant factors associated with persistent post-endodontic pain included prolonged preoperative pain, female gender, and previous chronic pain symptoms (Polycarpou, et al., 2005).
Two more recent reviews of chronic post-endodontic pain (Nixdorf, et al., 2010; Nixdorf, Moana-Filho, 2011) propose persistent dentoalveolar pain (PDAP) occurs in 1.6-3.6% of cases. The average pain reported as 7.2 on a visual analogue scale (where zero is no pain and 10 is pain as severe as it could be). The neuropathic pain was more common in middle-aged (mean 50.6 years of age) individuals with no sex predilection and occurred more frequently in the mandibular arch without any differences attributable to the number of canals treated. Also, the majority of subjects most frequently endorsed their pain experiences as abnormal sensitivity to touch and pain when the area is pressed or rubbed.
Persistent pain after routine surgery is an emerging entity; in a study of 221 patients with trigeminal nerve injuries related to dentistry, 70% are reported to have chronic neuropathic post-traumatic pain in patients attending a nerve injury clinic (Renton, Yilmaz, 2011). Another study confirmed this phenomenon in 91 patients (Benoliel, et al, 2012).
Neuropathic pain (NP) syndromes are chronic pain disorders that develop after a lesion of the peripheral nervous structures that are normally involved in signaling pain. Mechanisms for this process have been investigated but as yet elude us (Fried, et al., 2001; Benoliel, Kahn, Eliav, 2012; Forssell, et al., 2007). There are increasing reports of persistent pain post-dental surgery, likely to be related to post-traumatic neuropathy due to nerve injury (Queral-Godoy, et al., 2006; Rodriquez-Lozano, et al., 2010; Renton, et al., 2012). This increased recognition of chronic persistent pain after surgery is also occurring in general surgery (Kehlet, et al., 2007), reaffirming that a large proportion of patients attending chronic pain management clinics do, in fact, have post-surgical neuropathic pain.
There are relatively few reports of persistent pain subsequent to endodontic procedures (Nixdorf, et al., 2010). Persistent pain after endodontics has been reported to occur in 3%-13% of patients (Polycarpou, et al., 2005). Surgical endodontics are reported to result in chronic neuropathic pain in 5% of patients (Campbell, Parks, Dodds, 1990). Groltze, et al., reported on 11 patients with endodontic associated neuropathy and their management. They similarly reported that the neurological findings were dominated by hypesthesia and dysesthesia with 50% of patients reporting pain. Eleven of 61 patients with endodontic NI presented with significant pain, and only 40% responded to surgery (Pogrel, 2007). Ninety percent of the 10 patients in a later study reported chronic pain with 50% experiencing allodynia (pain evoked to non-noxious stimuli) and hyper-algesia (increased pain to noxious stimuli — for example, a pinprick or partners’ bristles on kissing).
The characteristics of NP differ substantially from those of other chronic pain states — i.e., chronic nociceptive pain, which develops while the nervous system that is involved in pain processing is intact. As well as the existence of negative somatosensory signs (deficit in function) there are other features that are characteristic of neuropathic conditions. Paresthesias are symptoms typically described by patients that are bothersome but not painful. Furthermore, NP states require different therapeutic approaches such as anticonvulsants, which are not effective in nociceptive pain, according to NICE guidance. Thus, symptoms experienced by patients with post-traumatic neuropathy of the trigeminal nerve can range from next to no symptoms, such as minimal anesthesia in a small area, to devastating effects on the patient’s quality of life (Renton, Yilmaz, 2011).
There is a lot of confusion in the terminology used for chronic pain related to surgical interventions. Chronic pain after surgery has too many names, including surgically induced neuropathic pain (SNIP), chronic post-surgical pain (CPSP), post-traumatic neuropathy (PTN), postoperative neuropathic pain (PPNP), and phantom limb pain.
There are two main types of chronic pain relating to surgery:
- Induced intractable neuropathic pain post-surgery (new or worsening pain)
- Persistent neuropathic pain pre- and post-surgery (persistent dento-alveolar pain [PDAP] or pre-existing neuropathic pain before the surgery).
The criteria for diagnosing CPSP (Macrae, 2008) include:
- Pain developed after surgery
- Minimum 2-month duration
- Other causes of pain have been excluded (infection, persistent malignancy, misdiagnosis)
- Excluded preoperative pain from other cause
For CPSP, a neuropathy does not have to be demonstrated in these cases, whereas if post-traumatic neuropathy is present, then a neuropathy will be present. However, other features of neuropathic post-surgical pain will be present, including:
- Pain
- Hyperesthesia
- Allodynia pain with non-noxious stimulus
- Pain on touch/cold/hot
- Hyperalgesia increased pain to painful stimulus
- Altered sensation
- Paresthesia — pins and needles, formication, many descriptions
- Dysesthesia — uncomfortable sensations, often burning
- Numbness — hypoesthesia
Chronic pain induced by surgery results in significant functional and psychological implications for the patients (Smith, et al., 2013; Renton, Yilmaz, 2011). Therefore, holistic assessment and management are imperative to manage these patients. Optimal management of chronic pain will help the patient move forward with support from Liaison Psychiatry Service (Renton, Yilmaz, 2012). Lifelong chronic pain is a likely outcome with endodontic-related IANIs and can mean that patients are on long-term medications. This reinforces the emphasis on prevention of these IANIs as they are so difficult to manage.

As a result of these severe consequences, complaints and litigation often ensue. In a review of 16 medicolegal claims related to persistent altered sensation following endodontic treatments, the typical profile of a claim was a female patient who underwent an endodontic treatment for a second mandibular molar, which was associated with overfilling. None of the claims were reported by the practitioners, and all cases were identified as a result of the patient’s demand for financial compensation, either directly or by legal actions (Givol, et al., 2011).
Mechanism of nerve injury related to endodontic treatment
All trauma-sensitive neural tissues will result in various neurophysiological effects as the IAN is contained within a bony canal, which predisposes it to compression and possible ischemic-type injury. Compression of peripheral sensory nerves over 6 hours can evoke nerve fiber atrophy (Shimpo, et al., 1987). Ischemia alone without direct nerve damage will cause sufficient neural inflammation and damage to cause permanent nerve injury (Park, Kim, Moon, 2012). Three months after the IAN injury, permanent central and peripheral changes occur within the nervous system subsequent to injury, which are unlikely to respond to surgical treatment intervention (Yekta, et al., 2010).
 IANIs related to RCTs can be due to local anesthesia, which is rare and usually associated with acute pain during block injections. IANIs related to endodontic treatment can be due to mechanical, chemical, and hemorrhagic insults. The injury may be to the nerve itself (extraneural or intraneural) of varying sites and/or associated vessels:
IANIs related to RCTs can be due to local anesthesia, which is rare and usually associated with acute pain during block injections. IANIs related to endodontic treatment can be due to mechanical, chemical, and hemorrhagic insults. The injury may be to the nerve itself (extraneural or intraneural) of varying sites and/or associated vessels:
- Mechanical direct reamer/indirect scarring — excessive root canal preparation often causes enlargement of the apical foramen and loss of constriction, which favors extravazation of irrigation products or filling material beyond the apex, which, in turn, may cause neural injury of chemical or mechanical origin. The diameter of bone lesions of endodontic origin may also influence the occurrence of paresthesia, especially when associated with the premolars and lower molars (Koseoglu, et al., 2006). Rowe (1983) argued that mechanical damage to the nerve caused by an endodontic instrument can be repaired during the healing process and that this form of paresthesia is generally temporary. Figures 2-4 are examples of post-endodontic radiographic overfills in our patient sample.
- Dental materials may exert damaging effects on nerve conduction as a result of their physical and chemical properties (Table 1).
- Even chemically bland materials such as gutta percha may cause irreversible neural injury following their entry to the inferior dental canal in a molten, thermoplastic state, partially resulting from direct thermal damage, and partly from nerve compression as the material cools and contracts (Fanibunda, Whitworth, Steele, 1998).
- Pressure ischemia from bleed or endodontic preparation.
- Periapical infection may also cause IANI (Morse, 1997). The possibility of paresthesia due to extrusion of microorganisms should be considered, given the potential for biological aggression, although this mechanism has not been explored in the literature (Table 1).
- Chemical toxicity and mechanical pressure produced by leakage of sealers into areas close to the mandibular canal are other potential mechanisms for nerve injury related to endodontic procedures, with the IAN and mental nerve being most affected. The materials most commonly associated with these complications are those containing paraformaldehyde (Ahonen, Tjaderhane, 2011). However, most endodontic materials have very high pHs resulting in immediate nerve damage likely to be permanent (Brodin, et al., 1982; Brodin, 1988; Serper, et al., 1998; Kozam, Newark, 1977).
- Hemaglobin irritates nerve tissue due to the iron content.
Risk factors
Tilotta-Yasukawa and colleagues (2006) determined the proximity of the apex of the premolars and molars in relation to the mandibular canal, as well as the relationship between the IAN and its corresponding artery, with the goal of understanding how endodontic filling material spreads through the bone to penetrate the mandibular canal.
They observed that the distance between the dental apex and the mandibular canal was more variable (and generally greater) for the first molar than for the second and third molars (1 mm-4 mm versus less than 1 mm [35 cases in 40 mandibles examined]).
The authors concluded that, in the posterior region, the mandibular bone is not very dense and has a greater amount of trabecular bone. In addition, the presence of numerous vacuoles in the mandibular bone in the molar region facilitates the spread of irrigation products and filling material toward the inferior alveolar neurovascular set (Knowles, Jergenson, Howard, 2003).
In a study of 135 patients with inferior alveolar nerve injuries caused by dental treatment or malignancy, 22% presented with dysesthesia that was significantly associated with the female gender and previous chronic pain (Oshima, et al., 2009).
GDP inadequacies
GDP inadequacies of GDP root canal treatments were highlighted by Jenkins, Hayes and Dummer (2001).
Chemical nerve injuries
Regarding chemical nerve injuries from leakage through the apex, such as sodium hydroxide — there are several reports of extreme pain and swelling resulting from endodontic irrigation with sodium hypo-
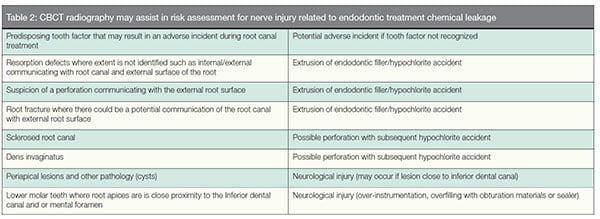
chlorite, with a multitude of associated complications including neuropathy. Kleier, Averbach, and Mehdipour (2008) surveyed 342 Diplomates of the American Board of Endodontics. Of the Diplomates that responded, 132 reported experiencing a sodium hypochlorite accident. The risk factors included women compared with men (p < 0.0001); maxillary teeth compared with mandibular teeth (p < 0.0001); posterior more than anterior teeth (p < 0.0001); a diagnosis of pulp necrosis with radiographic findings of periradicular radiolucency were positively associated with such accidents (p < 0.0001). Table 2 lists potential risk factors for sodium chlorite and other chemical leaks that may contribute to nerve injury.
Technical detectable overfill
Technical detectable overfill (Figures 2-3) occurred in 60% of cases and over-instrumentation during preparation. Any tooth requiring endodontic therapy, which is in close proximity to the IAN canal, should require special attention, including sodium hypochlorite (as irrigation) and calcium hydroxide (as sealant; calcium hydroxide medicaments breach the canal roof, precipitating a vascular bleed resulting in hemo-globin irritation of the nerve due to the iron content) (Escoda-Francoli, et al., 2007; Blanas, Kienle, Sándor, 2004).
Over-instrumentation (Figures 4-5)
The practitioner should be trained in root length assessment and root canal preparation. If the apex is proximal to the IAN canal, and if the canal is over-instrumented, there is increased risk of damage to the nerve. If the canal is overprepared and the apex opened, the nerve may be damaged by preparatory files, overfilling using pressurized thermal filling, and pressure and ischemia due to intracanal hemorrhage.
Dental factors
Many studies report the most likely teeth related to IANI are first molar and second premolars. Chikvashvili (2011) reported a significant correlation between the tooth location, and the suggested cause of nerve injury was found. Other dental factors can be found in Table 2.
 Periapical lesions (Figure 6)
Periapical lesions (Figure 6)
In one retrospective study, the incidence of mental paresthesia resulting from periapical infection or pathology was 0.96%. In another (0.24% of cases in the same study), mental paresthesia was a complication of root canal treatment (caused by severe overfill in one case and iatrogenic perforation of mechanical instrumentation through the root and into the mental nerve in the second case). Neuritis neuropathy may be induced by local inflammatory factors related to the periapical lesions and, if persistent, can cause permanent IAN neuropathy.
Many case reports that document neuropathies associated with apical periodontitis are scant, but usually involve premolars and sensory disturbance in the distribution of the mental nerve (von Ohle, ElAyouti, 2010; Shadmehr, Shekarchizade, 2015). There is every expectation that carefully conducted RCT that limits instruments and materials within the tooth, or indeed tooth extraction will allow symptoms to resolve (Ahonen, Tjäderhane, 2011).
Proximity
Proximity of the tooth to the IDC is a risk factor reported in all studies, including teeth apices close to the IDC and premolars adjacent to the mental loop (Scarano, et al., 2007; Köseoğlu, et al., 2006). Mandibular teeth proximal to the IAN canal may have the apex of the tooth and may be adjacent or intruding into the IDC canal, and any small degree of leakage or overfilling may compromise the IAN. Assessment of the proximity of the tooth apex to the IAN canal has become significantly improved with cone beam CT (CBCT) scanning with the attendant risk of additional radiation, and may not provide significantly more information than a plane long cone radiograph. Most CBCT assessment of tooth positioning in relation to the IAN canal is based on m3m prior to extraction (Tilotta-Yasukawa, et al., 2006).
It is a concern that some endodontic programs are training delegates to overfill past the apex by 1 mm-2 mm for optimal results, as there is insufficient evidence to support this recommendation and no mention of pre-surgical safety zone assessment to avoid nerve injuries in those teeth with apices close to the IDC.
The second part of this article will examine CBCT guidance in endodontic-related nerve injuries, as well as the diagnosis, assessment, and management in practice.
References
- Ahonen M, Tjäderhane L. Endodontic-related paresthesia: a case report and literature review. J Endod. 2011;37(10):1460-1464.
- Alves FR, Coutinho MS, Gonçalves LS. Endodontic-related facial paresthesia: systematic review. J Can Dent Assoc. 2014;80:e13
- Benoliel R, Kahn J, Eliav E. Peripheral painful traumatic trigeminal neuropathies. Oral Dis. 18(4):317-332.
- Benoliel R, Zadik Y, Eliav E, Sharav Y. Peripheral painful traumatic trigeminal neuropathy: clinical features in 91 cases and proposal of novel diagnostic criteria. J Orofac Pain. 2012;26(1):49-58.
- Blanas N, Kienle F, Sándor GK. Inferior alveolar nerve injury caused by thermoplastic gutta-percha overextension. J Can Dent Assoc. 2004;70(6):384-387.
- Brodin P. Neurotoxic and analgesic effects of root canal cements and pulp-protecting dental materials. Endod Dent Traumatol. 1988;4(1):1-11.
- Brodin P, Røed A, Aars H, Orstravik D. Neurotoxic effects of root filling materials on rat phrenic nerve in vitro. J Dent Res. 1982;61(8):1020-1023.
- Caissie R, Goulet J, Fortin M, Morielle D. Iatrogenic paresthesia in the third division of the trigeminal nerve: 12 years of clinical experience. J Can Dent Assoc. 2005;71(3):185-190.
- Campbell RL, Parks KW, Dodds RN. Chronic facial pain associated with endodontic therapy. Oral Surg Oral Med Oral Pathol. 1990;69(3):287-290.
- Chikvashvili J. Overcoming unforeseen incidents: what to do when an unlikely event occurs. Compend Contin Educ Dent. 2011;32(5):44-48.
- Dempf R, Hausamen JE. Lesions of the inferior alveolar nerve arising from endodontic treatment. Aust Endod J. 2000; 26(2):67-71.
- Escoda-Francoli J, Canalda-Sahli C, Soler A, Figueiredo R, Gay-Escoda C. Inferior alveolar nerve damage because of overextended endodontic material: a problem of sealer cement biocompatibility. J Endod. 2007;33(12):1484-1489.
- Fanibunda K, Whitworth J, Steele J. The management of thermomechanically compacted gutta- percha extrusion in the inferior dental canal. Br Dent J. 1998;184(7):330-332.
- Forman GH, Rood JP. Successful retrieval of endodontic material from the inferior alveolar nerve. J Dent. 1977;5(1):47-50.
- Forssell H, Tenovuo O, Silvoniemi P, Jääskeläinen SK. Differences and similarities between atypical facial pain and trigeminal neuropathic pain. Neurology. 2007; 69(14):1451-1459.
- Fried K, Bongenhielm U, Boissonade FM, Robinson PP. Nerve injury-induced pain in the trigeminal system. Neuroscientist. 2001;7(2):155-165.
- Gallas-Torreira MM, Reboiras-Lopez MD, Garcia-Garcia A, Gandara-Rey J. Mandibular nerve paresthesia caused by endodontic treatment. Med Oral. 2003;8(4):299-303.
- Gambarini G, Plotino G, Grande NM, et al. Differential diagnosis of endodontic-related inferior alveolar nerve paresthesia with cone beam computed tomography: a case report. Int Endod J. 2011; 44(2):176-81.
- Givol N, Rosen E, Bjørndal L, Taschieri S, Ofec R, Tsesis I. Medico-legal aspects of altered sensation following endodontic treatment: a retrospective case series. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112(1):126-131.
- González-Martin M, Torres-Lagares D, Gutierrez-Perez JL, Segura-Egea JJ. Inferior alveolar nerve paresthesia after overfilling of endodontic sealer into the mandibular canal. J Endod. 2010;36(8):1419-1421.
- Grötz KA, Al-Nawas B, de Aguiar EG, Schulz A, Wagner W. Treatment of injuries to the inferior alveolar nerve after endodontic procedures. Clin Oral Investig. 1998;2(2):73-76.
- Jenkins SM, Hayes SJ, Dummer PM. A study of endodontic treatment carried out in dental practice within the UK. Int Endod J. 2001;34(1):16-22.
- Klasser GD, Kugelmann AM, Villines D, Johnson BR. The prevalence of persistent pain after nonsurgical root canal treatment. Quintessence Int. 2011;42(3):259-269.
- Kleier DJ, Averbach RE, Mehdipour O. The sodium hypochlorite accident: experience of diplomates of the American Board of Endodontics. J Endod. 2008;34(11):1346-1350.
- Knowles KI, Jergenson MA, Howard JH. Paresthesia associated with endodontic treatment of mandibular premolars. J Endod. 2003;29(11):768-770.
- Köseoğlu BG, Tanrikulu S, Sübay RK, Sencer S. Anesthesia following overfilling of a root canal sealer into the mandibular canal: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(6):803-806.
- Kozam G. The effect of eugenol on nerve transmission. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1977;44(5):799-805.
- Lobb WK, Zakariasen KL, McGrath PJ. Endodontic treatment outcomes: do patients perceive problems? J Am Dent Assoc. 1996;127(5):597-600.
- Lopez-Lopez J, Estrugo-Devesa A, Jané-Salas E, Segura-Egea JJ. Inferior alveolar nerve injury resulting from overextension of an endodontic sealer: non-surgical management using the GABA analogue pregabalin. Int Endod J. 2012;45(1):98-104.
- Macrae WA. Chronic post-surgical pain: 10 years on. Br J Anaesth. 2008;101(1):77-86.
- Marbach JJ, Hulbrock J, Hohn C, Segal AG. Incidence of phantom tooth pain: an atypical facial neuralgia. Oral Surg Oral Med Oral Pathol. 1982;53(2):190-193.
- Marques TM, Gomes JM. Decompression of inferior alveolar nerve: case report. J Can Dent Assoc. 2011;77:b34.
- Morse DR. Infection-related mental and inferior alveolar nerve paresthesia: literature review and presentation of two cases. J Endod. 1997;23(7):457-460.
- Nixdorf DR, Moana-Filho EJ, Law AS, McGuire LA, Hodges JS, John MT. Frequency of persistent tooth pain after root canal therapy: a systemic review and meta-analysis. J Endod. 2010;36(2): 224-230.
- Nixdorf DR, Moana-Filho EJ. Persistent dento-alveolar pain disorder (PDAP): Working towards a better understanding. Rev Pain. 2011;5(4):18-27.
- Orr DL II. Paresthesia of the trigeminal nerve secondary to endodontic manipulation with N2. Headache. 1985;25(6):334-346.
- Oshima K, Ishii T, Ogura Y, Aoyama Y, Katsuumi I. Clinical investigation of patients who develop neuropathic tooth pain after endodontics procedures. J Endod. 2009;35(7):958-961.
- Park YT, Kim SG, Moon SY. Indirect compressive injury to the inferior alveolar nerve caused by dental implant placement. J Oral Maxillofac Surg. 2012;70(4):e258-e259.
- Pelka M, Petschelt A. Permanent mimic musculature and nerve damage caused by sodium hypochlorite: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106(3): e80-e83.
- Pogrel MA. Damage to the inferior alveolar nerve as the result of root canal therapy. J Am Dent Assoc. 2007;138(1):65-69.
- Polycarpou N, Ng YL, Canavan D, Moles DR, Gulabivala K. Prevalence of persistent pain after endodontic treatment and factors affecting its occurrence in cases with complete radiographic healing. Int Endod J. 2005;38(3):169-178.
- Poveda R, Bagán JV, Fernández JM, Sanchis JM. Mental nerve paresthesia associated with endodontic paste within the mandibular canal: report of a case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102(5):e46-e49.
- Qrstravik D, Brodin P, Aas E. Paraesthesia following endodontic treatment: survey of the literature and report of a case. Int Endod J. 1983;16(4):167-172.
- Queral-Godoy E, Vazquez-Delgado E, Okeson JP, Gay-Escoda C. Persistent idiopathic facial pain following dental implant placement: a case report. Int J Oral Maxillofac Implants. 2006;21(1):136-140.
- Renton T, Dawood A, Shah A, Searson L, Yilmaz Z. Post-implant neuropathy of the trigeminal nerve. A case series. Br Dent J. 2012;212(11):e17.
- Renton T, Yilmaz Z. Profiling of patients presenting with posttraumatic neuropathy of the trigeminal nerve. J Orofac Pain. 2011;25(4):333-344.
- Renton T, Yilmaz Z. Managing iatrogenic trigeminal nerve injury: a case series and review of the literature. Int J Oral Maxillofac Surg. 2012;41(5):629-637.
- Rodriquez-Lozano F, Sanchez-Pérez A, Moya-Villaescusa MJ, Rodriguez-Lozano A, Saez-Yuguero MR. Neuropathic orofacial pain after dental implant placement: review of the literature and case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(4):e8-e12.
- Rowe AH. Damage to the inferior dental nerve during or following endodontic treatment. Br Dent J. 1983; 155(9):306-307.
- Scarano A, Di Carlo F, Quaranta A, Piattelli A. Injury of the inferior alveolar nerve after overfilling of the root canal with endodontic cement: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104(1): e56-e59.
- Scolozzi P, Lombardi T, Jaques B. Successful inferior alveolar nerve decompression for dysesthesia following endodontic treatment: report of 4 cases treated by mandibular sagittal osteotomy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;97(5):625-631.
- Serper A, Uçer O, Onur R, Etikan I. Comparative neurotoxic effects of root canal materials on rat sciatic nerve. J Endod. 1998;24(9):592-594.
- Shadmehr E, Shekarchizade N. Endodontic periapical lesion-induced mental nerve paresthesia. Dent Res J (Isfahan). 2015;12(2):192-196.
- Shimpo T, Gilliatt RW, Kennett RP, Allen PJ. Susceptibility to pressure neuropathy distal to a constricting ligature in the guinea-pig. J Neurol Neurosurg Psychiatry. 1987;50(12):1625-1632.
- Smith JG, Elias LA, Yilmaz Z, et al. The psychosocial and affective burden of posttraumatic neuropathy following injuries to the trigeminal nerve. J Orofac Pain. 2013;27(4):293-303.
- Tilotta-Yasukawa F, Millot S, El Haddioui A, Bravetti P, Gaudy JF. Labiomandibular paresthesia caused by endodontic treatment: an anatomic and clinical study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102(4):e47-e59
- Vasilakis GJ, Vasilakis CM. Mandibular endodontic-related paresthesia. Gen Dent. 2004;52(4): 334-338.
- von Ohle C, ElAyouti A. Neurosensory impairment of the mental nerve as a sequel of periapical periodontitis: case report and review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;110(4) e84-e89.
- Yaltirik M, Ozbas H, Erisen R. Surgical management of overfilling of the root canal: a case report. Quintessence Int. 2002;3(9):670-672.
- Yekta SS, Smeets R, Stein JM, Ellrich J. Assessment of trigeminal nerve functions by quantitative sensory testing in patients and healthy volunteers. J Oral Maxillofac Surg. 2010;68(10):2437-2451.
- Zmener O. Mental nerve paresthesia associated with an adhesive resin restoration: a case report. J Endod. 2004;30(2):117-119.
Stay Relevant With Endodontic Practice US
Join our email list for CE courses and webinars, articles and more..


