Carolyn Primus, inventor of White ProRoot® MTA and dental researcher, offers insights on using bioactive materials and her life in endodontics.

Carolyn Primus, BS, MS, PhD, inventor, researcher, manufacturer, teacher, and humanitarian
What can you tell us about your background?
I earned a BS degree in Ceramic Engineering, then MS and PhD degrees in Materials Science and Engineering. Before and after getting my degrees, I performed research in national laboratories related first to nuclear weapons and naval defense, and then to consumer products. After that, there I began a 30-year career in dental materials research and development for endodontics, fixed prosthodontics, and orthodontics. I recently retired to write about bioceramics in dentistry.
What originally attracted you to the specialty of endodontics?
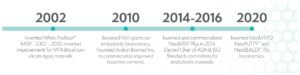
I was consulting for a leading dental products manufacturer that charged me with developing the manufacturing methods for the original hydraulic bioactive cement formula, commonly known as mineral trioxide aggregate (MTA). In 2002, I invented White ProRoot®* MTA.† Through the process of this work, I learned that hydraulic ceramic cements (MTA) offered immense potential for supporting healing of teeth.
From that realization, I began to think of new ideas, and applied to National Institutes of Health/National Institute of Dental and Craniofacial Research (NIH/NIDCR) for a Small Business Innovative Research (SBIR) grant. As part of the grant, I founded Avalon Biomed in 2010 to develop and market MTA Plus®, a line of affordable bioceramic dental medicaments for economical use worldwide. My vision was to develop a bio-active cement that had better properties than existing MTAs, with easier handling, faster setting, better color stability, and a price that endodontic, pediatric, and general dentists could afford.
Six years later I sold the assets of Avalon Biomed to NuSmile Ltd., the worldwide leader in pediatric esthetic restorative dentistry. Avalon Biomed is now a NuSmile subsidiary focused on endodontics. The past 4 years I have collaborated with NuSmile Ltd. on the innovation of three new products: NeoMTA®2, NeoPUTTY™, and NeoSEALER™ Flo.
What have been your proudest moments in terms of the clinical aspects of your life?
It’s a wonderful feeling when products come to fruition with test results and features that meet the needs of dentists and patients. I feel fortunate to have experienced this with several types of materials. I’m especially happy that NeoMTA Plus has been named the Top Bioactive Endodontic cement by DENTAL ADVISOR for the past 6 years. I’ve also had the good fortune of lecturing at dental schools and dental conferences on bioactive ceramics.
What prompted you to develop your newest products?
Endodontists have been vocal that the leading bioactive root and pulp products were too expensive, too time-consuming to mix, and prone to premature hardening in their packaging. Based on that information, we spent over 2 years developing three new products. NeoMTA 2 is an improved and economical powder-gel system with higher radiopacity, color stability, smooth consistency, washout resistance, and faster setting times. NeoPUTTY is an affordably priced, premixed, multi-indication pulp and root treatment paste in a syringe that virtually eliminates waste. NeoSEALER Flo is a bioactive bioceramic endodontic sealer in a syringe; its kit includes special tips that avoid waste and allow dispensing directly into a canal.
How challenging was this project?
Research and development of new product lines is always challenging, but NuSmile invested the financial and human resources in Avalon Biomed to ensure success. NuSmile doubled the bioceramic manufacturing area, purchased new capital equipment, and hired two bioengineers dedicated to product development. Expanded resources were applied to seek FDA clearance, Health Canada licenses, and CE certification. After extensive in vitro testing, several dental schools provided professional evaluations, including the College of Dentistry at the University of New England, the Dental College of Georgia at Augusta University, Texas A&M College of Dentistry, and the University of Texas School of Dentistry at Houston. A wide range of cutting-edge endodontists performed our ß-site testing of the products to verify customer satisfaction. Between NuSmile and our professional partners, a powerful team drove these projects to completion.
 What has been your biggest challenge in educating endodontists?
What has been your biggest challenge in educating endodontists?
I have been puzzled about several myths that persist about MTA bioactive cements. Many clinicians continue to assume that all MTA products are costly, slow-setting, “sandy,” and hard to handle. Many dentists remain unaware of how the bioactive bio-ceramics support healing of pulpal or periapical tissues for many procedures. Several bioactive ceramic cements containing bio-active calcium silicate cement are sporting similar product names and claims that confuse clinicians. The differences among these products — especially regarding cost per dose — are tremendous; dentists need to be smart shoppers! Also, “bioactivity” is loosely used in advertisements for supra-gingival cements without the histological proof of bioactive hydroxyapatite formation.
What advice would you give to budding endodontists?
Read the clinical literature critically. Study the Safety Data Sheets to compare products. Question manufacturers about claims and nomenclature, so you understand the materials or equipment. Many wonderful products are available, but not all products are equally useful; make sure you understand the claims and what you are purchasing. Dental shows are excellent venues to get answers, and I hope they will soon resume.
What is the future of endodontics?
Endodontics has a bright future. Regenerative endodontics using bioactive materials will become more common. Instrumentation will remove less root dentin. Equipment and irrigation methods will access the canal anatomy more completely to further improve treatment of pulp necrosis and irreversible pulpitis. Bioactive bioceramic cements and root canal sealers will be central to future improvements. Adult pulpotomies with bioactive bioceramics may become a more viable alternative to conventional root canal therapy.
What would you have become, had you not become a dental materials researcher?
Early in my career, I aspired to be an astronaut, but I eventually decided I wanted to develop materials for use in healthcare. I’ve greatly enjoyed being part of the dental community, even though I believe dentistry is underappreciated for its contributions to public health. I feel honored to have been able to interact with the best endodontists and dentists in the world.
What are your hobbies, and what do you do in your spare time?
I’m an enthusiastic birder. Even in retirement, I serve as chair of the ADA and ISO committees on endodontic materials.
* PROROOT®, DENTSPLY® and DENTSPLY SIRONA® are registered trademarks of DENTSPLY SIRONA Inc.
† Patented under US Patent No. 7,892,342 assigned to DENTSPLY International Inc.
Read more about Avalon Biomed, which was founded by Carolyn Primus, inventor of White ProRoot® MTA, here: https://endopracticeus.com/avalon-biomed/
Stay Relevant With Endodontic Practice US
Join our email list for CE courses and webinars, articles and more..

