 Dr. John Rhodes discusses treatment for a technically challenging condition
Dr. John Rhodes discusses treatment for a technically challenging condition
Introduction
This article describes the nonsurgical retreatment of a maxillary first molar (UL6) and the management of a pulp floor perforation.
History
A 52-year-old man was referred to an endodontic specialist following an attempt to retreat UL6. Unfortunately, a Gates Glidden bur had broken while attempting to remove gutta percha from the root canals and could not be retrieved.
A comprehensive medical history was uncomplicated.
Clinical examination
Intraoral examination revealed that UL6 was slightly tender to palpation.
The tooth was restorable and had a temporary (MO) compomer filling sealing the access.
There was no abnormal mobility, no significant or increased periodontal pocketing, or bleeding on probing.
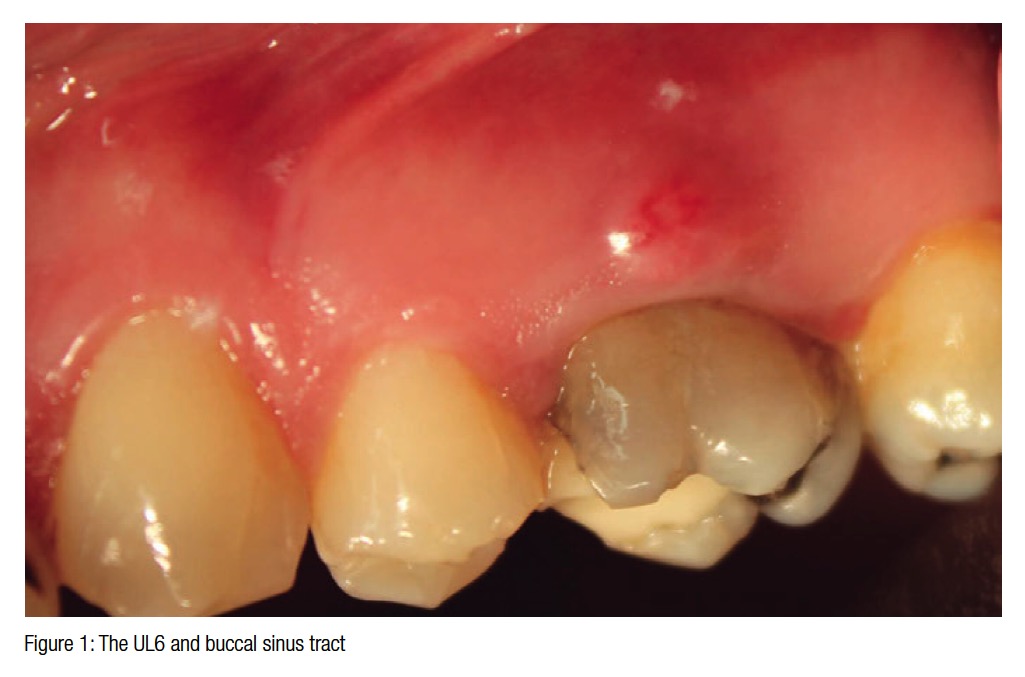
Soft tissue examination revealed that there was a buccal sinus tract through which pus could be expressed.
Sensitivity testing with Hygenic® Endo Ice® (Coltene) showed that the adjacent teeth were vital.
A paralleling periapical radiograph of the UL6 was exposed using a Rinn Holder (Rinn, Dentsply ).
The radiograph showed good marginal bone levels. The UL5 and UL7 were minimally restored with no evidence of periapical pathology, and the root canals appeared sclerosed. An unerupted UL8 was present.
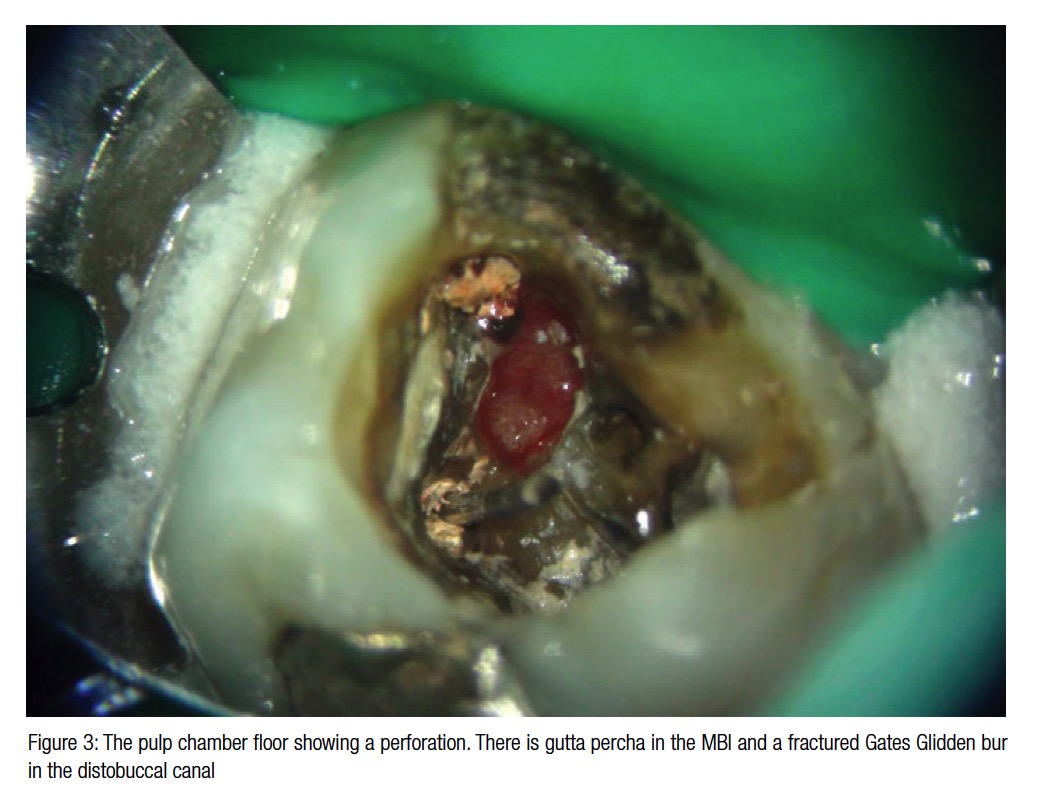
The UL6 had been root filled with gutta percha, but the canals were under-prepared and under-filled. The second mesiobuccal canal (MBII) had been located. A fractured Gates Glidden bur was lodged in the distobuccal canal. The inferior border of the maxillary antrum was visible and reached its lowest point between the first and second molars. There was potential radiolucency in the trifurcation and periapically around the MB root; however, this was masked by the proximity of the antrum.
 Diagnosis
Diagnosis
Failed root filling and chronic periapical (periradicular) abscess.
The root canal filling had most probably failed as a result of persistent bacterial infection in the root canal.
Treatment options
- Root canal retreatment and restoration with a cusp coverage onlay or crown
- Extraction and nothing
- Extraction and replacement with an implant
The prognosis for root canal retreatment should be good, and the patient could expect the tooth to remain functional for many years.
Replacement with an implant may be feasible, but generally, well root-filled and restored teeth appear to function comparably to single tooth implants (Hannahan and Eleazer, 2008; Torebinejad, et al., 2007). Restored natural teeth also tend to be less costly to repair if and when they fail (Pennington, et al., 2009).
 After discussing all the treatment options, the patient elected to have the UL6 retreated nonsurgically.
After discussing all the treatment options, the patient elected to have the UL6 retreated nonsurgically.
Treatment
A two-visit strategy was adopted in this case to ensure adequate disinfection of the root canals, allow optimal management of a pulp floor perforation, and confirm healing of the sinus tract before obturation.
Profound anesthesia was provided by buccal and palatal infiltration of 4% articaine 1:100,000 adrenaline (Septodont).
Single tooth isolation with a latex rubber dam and a number 14 clamp provided a controlled operating field.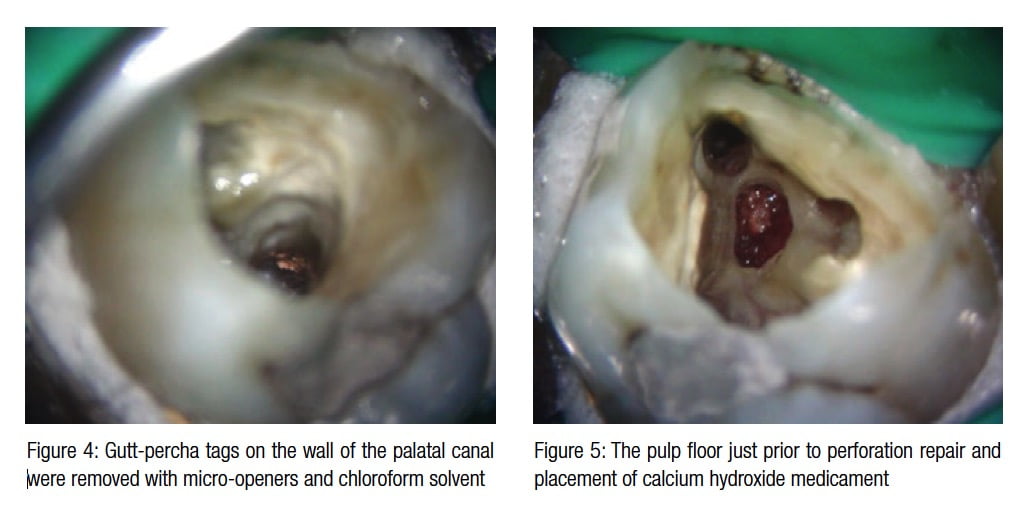
Using an operating microscope, the existing compomer filling was removed with a long-tapered diamond bur, and the margins of the cavity refined.
A cotton wool pellet was removed from the access cavity, and after rinsing with 3% sodium hypochlorite, a preliminary survey of the pulp floor could be carried out under low magnification with the operating microscope. This showed the fractured Gates Glidden bur seated in the distobuccal canal and a moderately large perforation adjacent to the mesiobuccal canal, extending into the furcation. Granulation tissue was visible through the perforation.
Gates Glidden burs provide a very efficient means of removing gutta percha and have a fail-safe stress point at the end of the shaft so that if they break, retrieval is easier. If they fracture when embedded in gutta percha, this cools and hardens around the bullet-shaped tip making them more difficult to remove. A simple technique to overcome this involves vibrating the fractured piece with a dry ultrasonic tip; the energy is dissipated as heat, re-plasticizing the gutta percha and allowing removal with Stieglitz forceps.
In this case, the fragment of a Gates Glidden bur was vibrated with a Start-X™ tip No. 3 (Dentsply).
The remaining bulk of gutta percha in all canals was rapidly removed with a Gates Glidden size 2 and Hedstroem file size 30 (Dentsply).
Straight-line access to all canals was then refined.
Working length estimations were made with an apex locator (Endo Analyzer, Sybron) and size 10 FlexoFile® (Dentsply Maillefer). A steady zero reading was achieved in all canals despite the perforation.
Patency was confirmed and glide paths created in the primary root canals with a ProGlider™ rotary instrument (Dentsply Maillefer; Dentsply Tulsa Dental Specialties), and these were then rapidly tapered using reciprocating WaveOne® primary and large instruments (Dentsply Maillefer; Dentsply Tulsa Dental Specialties). The canals were kept flooded with sodium hypochlorite and patency recapitulated throughout preparation.
The root canals were inspected under high magnification, and any tags of gutta percha on the canal walls removed with an Endo-Explorer (Dentsply Maillefer; Hu-Friedy). A small amount of chloroform was used as a solvent to dissolve any gutta percha lodged in lateral anatomy; this was wicked with sterile paper points.
The root canal system was disinfected with a solution of heated 3% sodium hypochlorite (Teepol) irrigant, delivered using a safe-ended needle (Henry Schein®). The needle was pre-bent and kept moving in the root canal to prevent extrusion of irrigant.
Disinfection was carried out over approximately 30 minutes. The sodium hypochlorite was agitated with a size 20 Irrisafe™ ultrasonic tip (Satelec Acteon) and an EndoActivator® (Dentsply Tulsa Dental Specialties) with a red tip. The solution was replenished every 2 minutes. A final sequence of 40% citric acid (Cerkamed) and 3% sodium hypochlorite completed irrigation.
The sodium hypochlorite solution was able to penetrate the perforation site but was not injected into it.
 The canals were dried with sterile paper points (Dentsply) and non-setting calcium hydroxide placed (Calasept®). Collagen matrix (Hemocollagene, Septodont) was packed through the perforation site to form a matrix onto which Biodentine® (Septodont) was compacted. Once set, the access was sealed with Cavit™ (3M ESPE) and Fuji IX® (GC).
The canals were dried with sterile paper points (Dentsply) and non-setting calcium hydroxide placed (Calasept®). Collagen matrix (Hemocollagene, Septodont) was packed through the perforation site to form a matrix onto which Biodentine® (Septodont) was compacted. Once set, the access was sealed with Cavit™ (3M ESPE) and Fuji IX® (GC).
One week later, the sinus had healed, the tooth was symptom-free, the canals dry, the integrity of the Biodentine confirmed, and the tooth ready for obturation.
 Calcium hydroxide inter-appointment medicament was removed with alternate citric acid and sodium hypochlorite irrigation. The canals were then dried and obturated with vertically compacted thermoplasticized gutta percha and AHPlus® sealer (Dentsply)
Calcium hydroxide inter-appointment medicament was removed with alternate citric acid and sodium hypochlorite irrigation. The canals were then dried and obturated with vertically compacted thermoplasticized gutta percha and AHPlus® sealer (Dentsply)
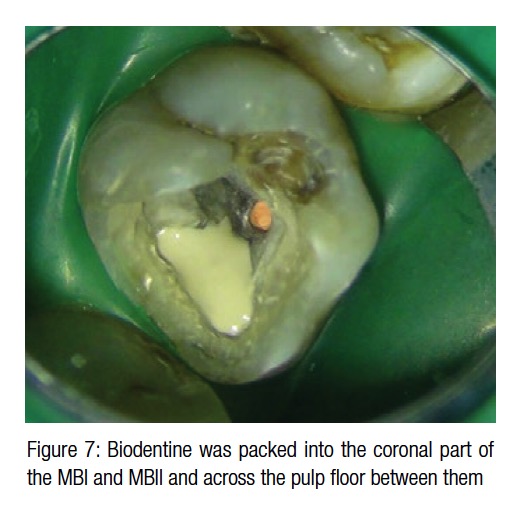
Biodentine was packed into the coronal 3 mm-4 mm of the MBI and MBII canals to ensure that the perforation site was well sealed. (Should the MB root ever need to be resected, this material would provide a good seal.)
Once the Biodentine had set, Fuji IX was used to seal the pulp floor. A Palodent®Plus sectional matrix (Dentsply) was placed and the cavity thoroughly washed. The cavity was selectively etched with 30% phosphoric acid and Scotchbond™ (3M ESPE) dual-cure bonding agent applied.
 A composite core was constructed with flowable bulk-fill composite (Smart Dentin replacement [SDR®] Dentsply) and Ceram-X® mono.
A composite core was constructed with flowable bulk-fill composite (Smart Dentin replacement [SDR®] Dentsply) and Ceram-X® mono.
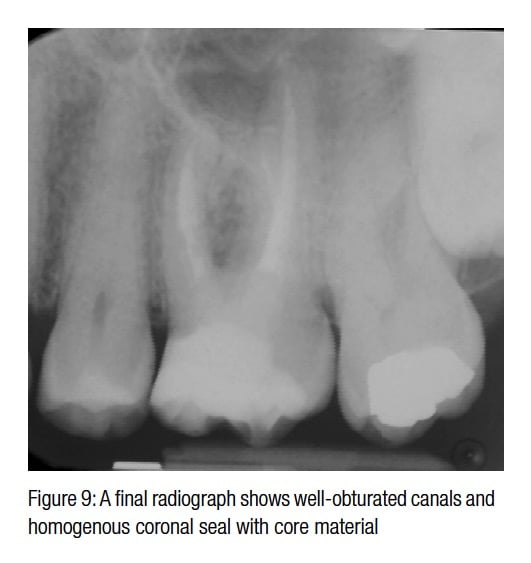
A final paralleling radiograph was exposed using a Rinn Holder. This showed a well-oburated root canal system and homogenous coronal seal with no voids.
A cusp coverage restoration was to be provided by the referring dentist to help prevent fracture.
Reviews will be carried out at 1 year and subsequently at 2 years, and 4 years if required in line with the “Quality guidelines for endodontic treatment: consensus report of the European Society of Endodontology” (2006).
Discussion
Iatrogenic pulp floor perforation can occur if the operator becomes disorientated when trying to locate canal orifices. The importance of using illumination and magnification during endodontic procedures cannot be overemphasized (Buhrley, et al., 2002; Schwarze, et al., 2002).
 Perforation repair can be technically challenging, and offering referral if treatment is beyond the expertise of the operator is good practice (General Dental Council Standards for the Dental Team 2013).
Perforation repair can be technically challenging, and offering referral if treatment is beyond the expertise of the operator is good practice (General Dental Council Standards for the Dental Team 2013).
Management of iatrogenic perforation is dependent on several factors (Fuss and Trope, 1996).
Level
There is a high risk of microleakage when perforation occurs at crestal bone level. Obtaining adequate coronal seal can be difficult, and direct communication with the oral cavity via the periodontal tissues means the prognosis may be guarded. If bone is present on the external aspect of the perforation, repair is normally feasible.
Location
Perforations that have damaged the root canal orifice can be more difficult to seal. In this case, there was dentin around the periphery of the MB canal, and the perforation was positioned away from any isthmus between the MBI and MBII.
Size
The larger the perforation, the greater the surface area that will need to be sealed.
Time
It is preferable to seal a perforation as soon as possible to prevent microleakage and bacterial contamination.
During retreatment, the existing material, any obstructions, and missed canals should be prepared and disinfected as efficiently as possible and at the first visit (Van Nieuwenhuysen, 1994).
When retreating teeth that have been obturated with gutta percha, most of the filling material can be removed mechanically. Gates Glidden burs are extremely efficient, and once a glide path has been established, tapering the canal with NiTi instruments will remove most of the remainder. Material lodged in fins and isthmuses will need to be removed using a solvent (Ferreira JJ, Rhodes JS, and Pitt Ford TR, 2001).
Mechanical preparation will reduce the bacterial load in an infected root canal (Byström and Sundqvist, 1981), but sodium hypochlorite in a concentration of at least 1% is required to kill the majority of bacteria in a contaminated canal system (Byström and Sundqvist, 1983). Bacteria aggregate as biofilms on the root canal wall. These bacteria are surrounded by matrix and can be difficult to remove.
Irrigants used during endodontic treatment, therefore, have to be agitated to disrupt biofilm (Ahmad M, Pitt Ford TJ, Crum LA, 1987). Ultrasonic activation removes more debris form the root canal than syringe irrigation alone (Burleson, et al., 2007).
A solution of 17% EDTA or citric acid as a final rinse (Byström and Sundqvist, 1985) is used to remove smear and has a positive benefit on outcome in retreatment cases (Ng, et al., 2011).
The concept of a two-visit strategy using a chemo-mechanical approach has been adopted since the 1980s. Dressing the canals (after preparation and disinfection) with calcium hydroxide for 7 days was shown to be effective at producing bacteria-free canals (Byström, Claesson, Sundqvist, 1985; Sjögren, et al., 1991). Universal use of a two-visit approach has been questioned as calcium hydroxide and sterile saline slurry limits but does not totally prevent regrowth of endodontic bacteria (Peters 2002). A systematic review of the literature reported that multiple visits with calcium hydroxide treatment did not improve clinical outcome, and there was a minimal level of evidence for considering one versus two appointments in nonsurgical endodontics (Hargreaves 2006).
Many endodontists have therefore adopted a single-visit approach for many cases with no significant postoperative difficulties nor reduced prognosis (Ng, et al., 2011).
Contrary to this, a two-visit strategy was adopted in this case to allow setting of the perforation repair material and confirm healing of the sinus tract before obturation.
Calcium silicate cements such as Biodentine and MTA have excellent sealing abilities and have many reported uses in endodontics (Parirokh and Torabinejad, 2010). The excellent performance of MTA in surgical endodontics is well published (Chong BS, Pitt Ford TR, and Hudson MB, 2003). Long-term follow-up case studies have demonstrated that MTA can be predictably used as a repair material for furcal perforation (Pace R, Giuliani V, Pagavino G, 2008).
Biodentine contains tricalcium silicate with additives such as powder and a liquid containing calcium chloride to speed up setting. Calcium silicate materials have excellent biocompatibility and are able to induce calcium-phosphate precipitation at the periodontal ligament interface allowing bony healing (Tay, et al., 2007; Torabinejad and Parirokh, 2010). With a reduced setting time compared to MTA, Biodentine is perhaps more user-friendly for perforation repair (Wongkornchaowalit N, Lertchirakarn V, 2011).
Definitive restoration of root-filled molar teeth with loss of marginal ridges should encompass cusp coverage to reduce the risk of tooth fracture under masticatory forces. (Whitworth JM, Walls AWG, and Wassell RW, 2002; Sorensen JA and Martinoff JT, 1984). In this case, the final restoration was to be completed by the referring dentist.
Conclusion
This article highlights the diagnosis and management of nonsurgical retreatment, instrument removal, and perforation repair with micro-endodontic techniques and Biodentine.
References
- Ahmad M, Pitt Ford TJ, Crum LA. Ultrasonic debridement of root canals: acoustic streaming and its possible role. J Endod. 1987;13(10):490-499.
- Buhrley LJ, Barrows MJ, BeGole EA, Wenckus CS. Effect of magnification on locating the MB2 canal in maxillary molars. J Endod. 2002;28(4):324-327.
- Byström A, Sundqvist G. Bacteriologic evaluation of the efficacy of mechanical root canal instrumentation in endodontic therapy. Scand J Dent Res. 1981;89(4):321-328.
- Byström A, Sundqvist G. Bacteriologic evaluation of the effect of 0.5 percent sodium hypochlorite in endodontic therapy. Oral Surg Oral Med Oral Pathol. 1983;55(3):307-312.
- Byström A, Sundqvist G. The antibacterial action of sodium hypochlorite and EDTA in 60 cases of endodontic therapy. Int Endod J. 1985;18(1):35-40.
- Byström A, Claesson R, Sundqvist G. The antibacterial effect of camphorated paramonochlorophenol, camphorated phenol and calcium hydroxide in the treatment of infected root canals. Endod Dent Traumatol. 1985;1(5):170-175.
- Burleson A, Nusstein J, Reader A, Beck M. The in vivo evaluation of hand / rotary / ultrasound instrumentation in necrotic human molars. J Endod. 2007;33(7):782-787
- Chong BS, Pitt Ford TR, Hudson MB. A prospective clinical study of Mineral Trioxide Aggregate and IRM when used as root- end filling materials in endodontic surgery. Int Endod J. 2003;36(8):520-526.
- Ferreira JJ, Rhodes JS, Pitt Ford TR. The efficacy of gutta-percha removal using ProFiles. Int Endod J. 2001;34(4):267–74.
- Fuss Z, Trope M. Root perforations: classification and treatment choices based on prognostic factors. Endod Dent Traumatol. 1996;12(6):255-264.
- General Dental Council. Standards for the dental team. September 2013. https://www.gdc-uk.org/Dentalprofessionals/Standards/Pages/standards.aspx. Accessed June 26, 2015.
- Hannahan JP and Eleazer PD. Comparison of success of implants versus endodontically treated teeth. J Endod. 2008;34(11):1302–1305.
- Hargreaves KM. Single-visit more effective than multiple-visit root canal treatment? Evid Based Dent. 2006;7(1):13–14.
- Ng YL, Mann V, Gulabivala K. A prospective study of the factors affecting outcomes of nonsurgical root canal treatment: part 1: periapical health. Int Endod J. 2011;44(7):583-609.
- Pace R, Giuliani V, Pagavino G. Mineral trioxide aggregate as a repair material for furcal perforation: case series. J Endod. 2008;34(9):1130-1133.
- Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review – part III: clinical applications, drawbacks, and mechanism of action. J Endod. 2010;36(3):400-413.
- Pennington MW, Vernazza CR, Shackley P, Armstrong NT, Whitworth JM, Steele JG. Evaluation of the cost-effectiveness of root canal treatment using conventional approaches versus replacement with an implant. Int Endod J. 2009;42(10):874-883.
- Peters LB, van Winkelhoff AJ, Buijs JF, Wesselink PR. Effects of instrumentation, irrigation and dressing with calcium hydroxide on infection in pulpless teeth with periapical bone lesions. Int Endod J. 2002;35(1):13-21.
- European Society of Endodontology. Quality guidelines for endodontic treatment: consensus report of the European Society of Endodontology. Int Endod J. 2006;39(12): 921–930.
- Schwarze T, Baethge C, Stecher T, Geurtsen W. Identification of second canals in the mesiobuccal root of maxillary first and second molars using magnifying loupes or an operating microscope. Aust Endod J. 2002;28(2):57-60.
- Sjögren U, Figdor D, Spångberg L, Sundqvist G. The antimicrobial effect of calcium hydroxide as a short-term intracanal dressing. Int Endod J. 1991;24(3):119–125.
- Sorensen JA, Martinoff JT. Intracoronal reinforcement and coronal coverage: a study of endodontically treated teeth. J Prosthet Dent. 1984;51(6):780-784.
- Tay FR, Pashley DH, Rueggeberg FA, Loushine RJ, Weller RN. Calcium phosphate phase transformation produced by the interaction of the Portland cement component of white mineral trioxide aggregate with a phosphate-containing fluid. J Endod. 2007;33(11):1347-1351.
- Torabinejad M, Parirokh M. Mineral trioxide aggregate: a comprehensive literature review – part II: Leakage and biocompatibility investigations. J Endod. 2010;36(2):190-202.
- Torabinejad M, Anderson P, Bader J, Brown LJ, Chen LH, Goodacre CJ, Kattadiyil MT, Kutsenko D, Lozada J, Patel R, Petersen F, Puterman I, White SN. Outcomes of root canal treatment and restoration, implant-supported single crowns, fixed partial dentures, and extraction without replacement: a systematic review. J Prosthet Dent. 2007;98(4):285-311.
- Van Nieuwenhuysen JP, Aouar M, D’Hoore W. Retreatment or radiographic monitoring in endodontics. Int Endod J. 1994;27(2):75-81.
- Whitworth JM, Walls AW, Wassell RW. Crowns and extra-coronal restorations: endodontic considerations: the pulp, the root-treated tooth and the crown. Br Dent J. 2002;192(6):315-20, 323-327.
- Wongkornchaowalit N, Lertchirakarn V. Setting time and flowability of accelerated Portland cement mixed with polycarboxylate superplasticizer. Journal of Endodontics. 2011;37(3):387-389.
Stay Relevant With Endodontic Practice US
Join our email list for CE courses and webinars, articles and more..


